| |
 |
Prof. Dr. Leonardo S. Santos is a Full Professor at Talca University. He is an internationally recognized scientist with expertise spanning the fields of Organic Chemistry, Mass Spectrometry, Agricultural, including omics technology, nanomedicine, drug discovery, and new analytical clinical technologies. He was formerly Director of Nanobiotechnology division of the Fraunhofer Chile Research (2010–2017), and Doctorate Head of the Institute of Chemistry of Natural Research at Talca University, Chile (2012–2023).
|
Prof. Dr. Santos established and built the Laboratory of Asymmetric Synthesis (2006), the Center for Nanoscience (CeNS, 2010), as well as the Laboratory of Nanotechnology and Mass Spectrometry (2011) in Talca. Based on these achievements, he was awarded the title Full Professor by Talca University in 2015. He has given more than 50 scientific presentations around the world, and has published more than 150 peer-reviewed scientific publications (h-index = 40) as well as one book. He has 5 pending patent applications and 4 granted patents. He has graduated more than 20 Bachelor’s students and 22 PhD students. At the managerial level, Prof. Dr. Santos has been extensively immersed in project acquisition and management, human resources, operations, technology and information management, product development, IP filing, multiple clinical trials, and engagement with regulatory bodies. He has helped to establish start-up companies, assisted in bringing 2 products to the market, and has served on multiple national and international advisory boards. He has consulted for the Chilean federal government and several Members of the Chilean Parliament, and has served on multiple scientific program advisory boards and working groups in academia and industry.
His research targets are selected on the basis of novel molecular architecture, important biological activity and interesting mechanism of action. Merging natural products chemistry, chemical synthesis and mechanisms using ESI-MS, these principles form the foundation for Dr. Santos’ research and educational programs. Furthermore, he continues to develop new methodologies based on new catalysts and the study of mechanistic pathways involving organometallic compounds through Mass Spectrometry experiments.
It is my conviction that Chemistry is, to a great extent, about chemical reactions - developing them, understanding them, and using them to make interesting molecules. Much of the activity in my research is directed at discovering new ways to make innovative chemistry. This includes (a) the design of unique strategies to particular families of structurally-intricate molecules, often possessing useful pharmacological properties, or (b) the invention of new reactions, or the development of useful aspects of known reactions, particularly their asymmetric variants, and finally (c) the development of new catalysts and the study of mechanistic pathways involving organometallic compounds through MALDI-MS and ESI-MS experiments. Man's fascination with natural substances goes back to ancient times. With the discovery of salicin from willow tree extracts and the development of aspirin in 1899, the art of exploiting natural products became a molecular science. The discovery of penicillin in 1928 and its subsequent development as an anti-infective agent represents another breakthrough in the history of natural products, and marked the beginning of a new era in drug discovery, in which bacteria and fungi were added to the plant kingdom as sources for biologically active compounds. Today, with marine organisms and other living creatures as additional sources of active compounds, the chemistry and biology of natural products represents a major avenue to drug discovery and development. Indeed, a large portion of today's major drugs have their origins in nature. It is, therefore, not surprising that one of the most flourishing and rewarding frontiers in modern science is the study of the chemistry and biology of natural products. But man's imagination does not stop at the frontiers defined by nature. With the ever-increasing power of organic synthesis, the synthetic organic chemist is poised to make important contributions by inventing and developing new enabling technologies for the generation of isolated natural products, but also of designed small organic molecules of broad structural diversity for binding to and modulating the function of biological targets.
In our laboratory, in pursuing the total synthesis of natural products, new synthetic strategies and methods are sought to solve the problems at hand, but also to remain as enabling technologies for chemistry, biology and medicine. Targets are selected on the basis of novel molecular architecture, important biological activity and interesting mechanism of action. Merging natural products chemistry, chemical synthesis and chemical biology, these principles form the foundation for our research and educational programs.
With regard to strategies, we have devised concise new routes to several families of natural products. Our objective is devise short, high yielding syntheses, through strategies that examine interesting aspects of structure and reactivity in an asymmetric way.

We have been actively involved in the development and study of useful synthetic methods. Electrospray ionization mass spectrometry (ESI-MS) is rapidly becoming an important technique for mechanistic studies of chemical reactions in solution including homogeneously catalyzed reactions and high-throughput screening of homogeneous catalysts.
Lewis Acid Enhanced Ethene Dimerization and Alkene Isomerization — ESI-MS Identification of Acitve Species
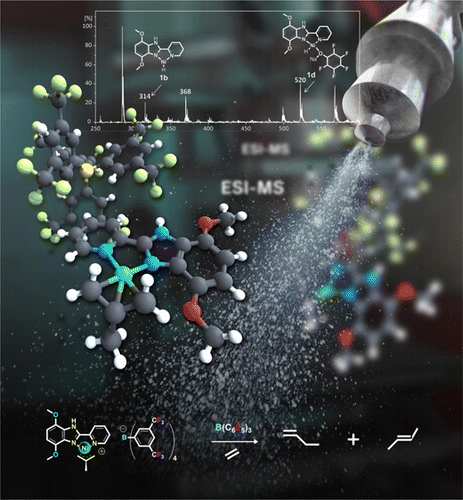
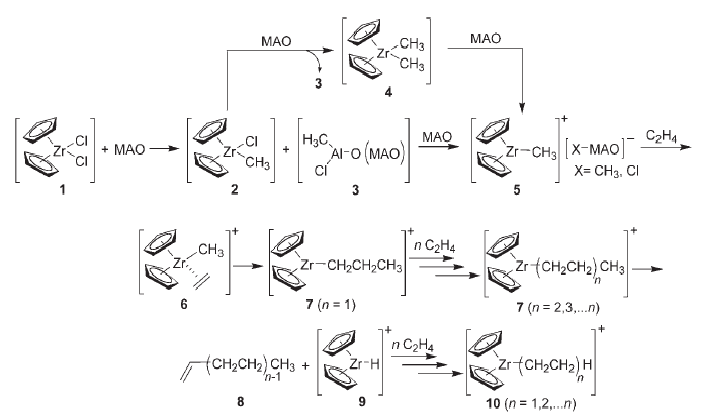
Probing the Mechanism of Ziegler-Natta Polymerization by On-Line Monitoring of Reaction through ESI-MS
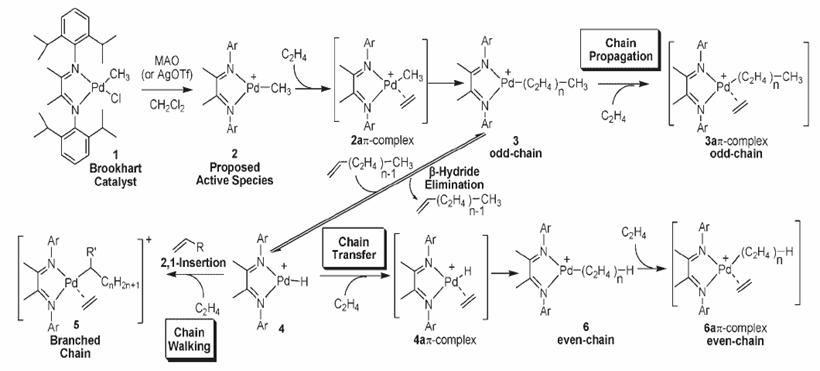
On-line Monitoring of Brookhart Polymerization Reactions by ESI-MS
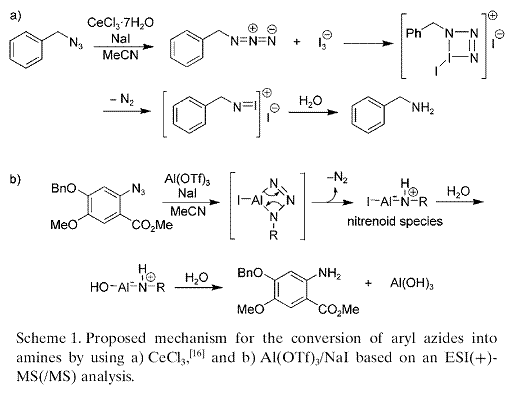
Chemoselective Aromatic Azido Reduction with Concomitant Aliphatic Azide Employing Al/Gd Triflates/NaI
Using APCI-MS(/MS) we have intercepted, characterized and proved the mechanism by Ionic Liquid species are vaporize at ambient conditions from heated (100-600 °C) solvent droplets. No evidence for vaporization of ILs as single neutral carbenes (for Im+) or AH species was observed.
Probing The Mechanism of Ionic Liquids distillation by APCI-MS
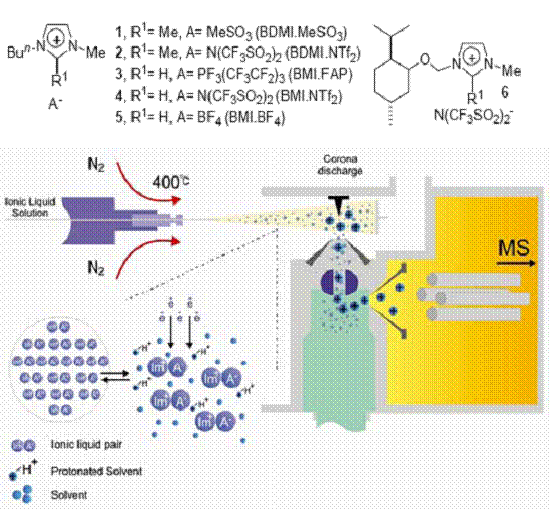
We are also actively pursuing the design of new methods for asymmetric synthesis based upon ruthenium, rhodium, palladium and titanium organometallics compounds. Finally, we have begun a program aimed at the development of new catalysts for organic synthesis.
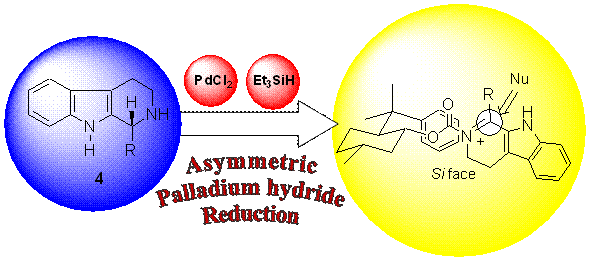
First example of bioreduction of beta-carboline imines
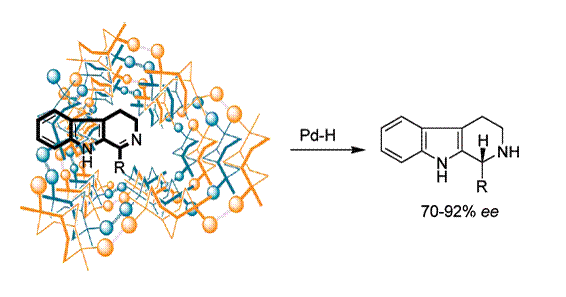
Palladium Asymmetric Reduction of b-Carboline Imines Mediated by Chiral Auxiliaries
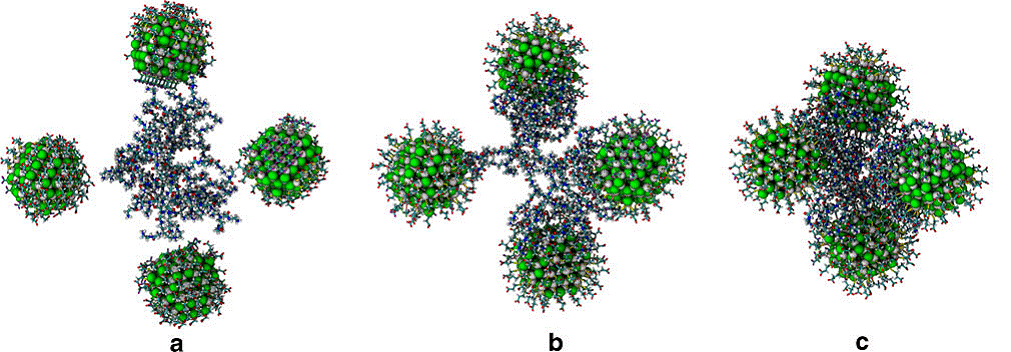
Schematic uptake of functionalized QDs/PAMAM supramolecular complexes by folate receptors in the phospholipidic bilayer of cancer cells. PAMAM dendrimer was functionalized using folate molecules in order to promote selective cellular uptake and imaging specificity.
QDs/PAMAM-FA nanocomposite
|
|
